
CDSCO Circular (23–24 February 2026)
Key highlights from the CDSCO circular on “Testing of drugs for grant of permissions for import/manufacture of New Drug for…
Read More
Key highlights from the CDSCO circular on “Testing of drugs for grant of permissions for import/manufacture of New Drug for…
Read More
G.S.R. 135(E) – Changes to the Drugs Rules On February 16, 2026, the Central Drugs Standard Control Organization (CDSCO) rolled…
Read More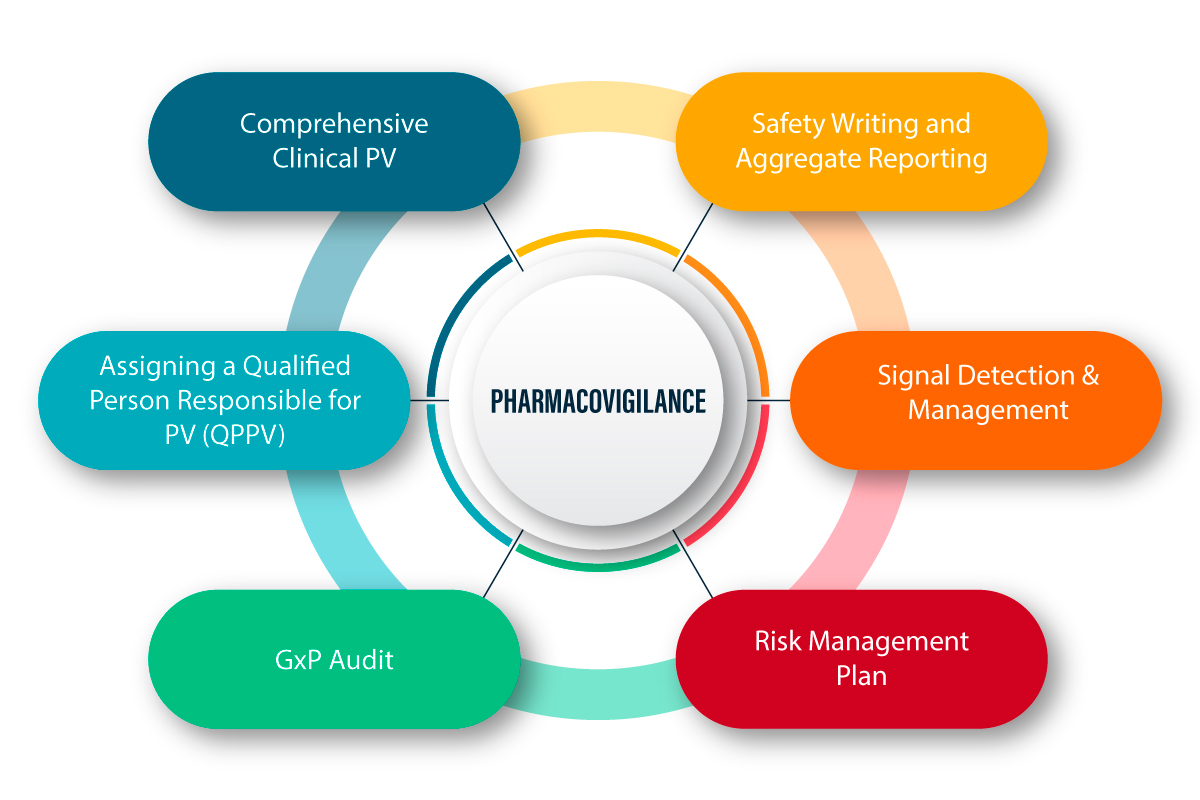
Let’s be real: in today’s business world, you can’t just ignore compliance. It’s not a box to tick—it’s something you’ve…
Read MoreHighlights and benefits of the recent U.S.–India trade agreement, with a focus on what it does (including tariff reductions) and…
Read MoreIndia-EU Free Trade Agreement — The “Mother of All Deals” Big news on the trade front: On January 27, 2026,…
Read More
Central Drugs Registration in India Accredited Consultants Pvt. Ltd., Your Regulatory Compliance Partner The Central Drugs Standard Control Organization (CDSCO)…
Read More
Diagnostic Registration in India Accredited Consultants Pvt. Ltd., Simplifying Regulatory Approvals In India, diagnostic kits, reagents, and in-vitro diagnostic (IVD)…
Read More
New Drug and Medical Registration in India ACPL Company, Your Trusted Regulatory Partner Bringing a new drug or medical device…
Read MoreBest Pharmacovigilance Services Provider in Noida End-to-End Drug Safety and Compliance Solutions We at ACPL provide exhaustive Pharmacovigilance (PV) services…
Read More
Best Pharmacovigilance Services Provider in India End-to-End Drug Safety and Compliance Solutions We at ACPL provide exhaustive Pharmacovigilance (PV) services…
Read More