
CDSCO Circular (23–24 February 2026)
Key highlights from the **CDSCO circular on “Testing of drugs for grant of permissions for import/manufacture of New Drug for…
Read MoreRegulatory updates, compliance insights, and industry analysis from the ACPL team.

Key highlights from the **CDSCO circular on “Testing of drugs for grant of permissions for import/manufacture of New Drug for…
Read More
G.S.R. 135(E) – Changes to the Drugs Rules On February 16, 2026, the Central Drugs Standard Control Organization (CDSCO) rolled…
Read More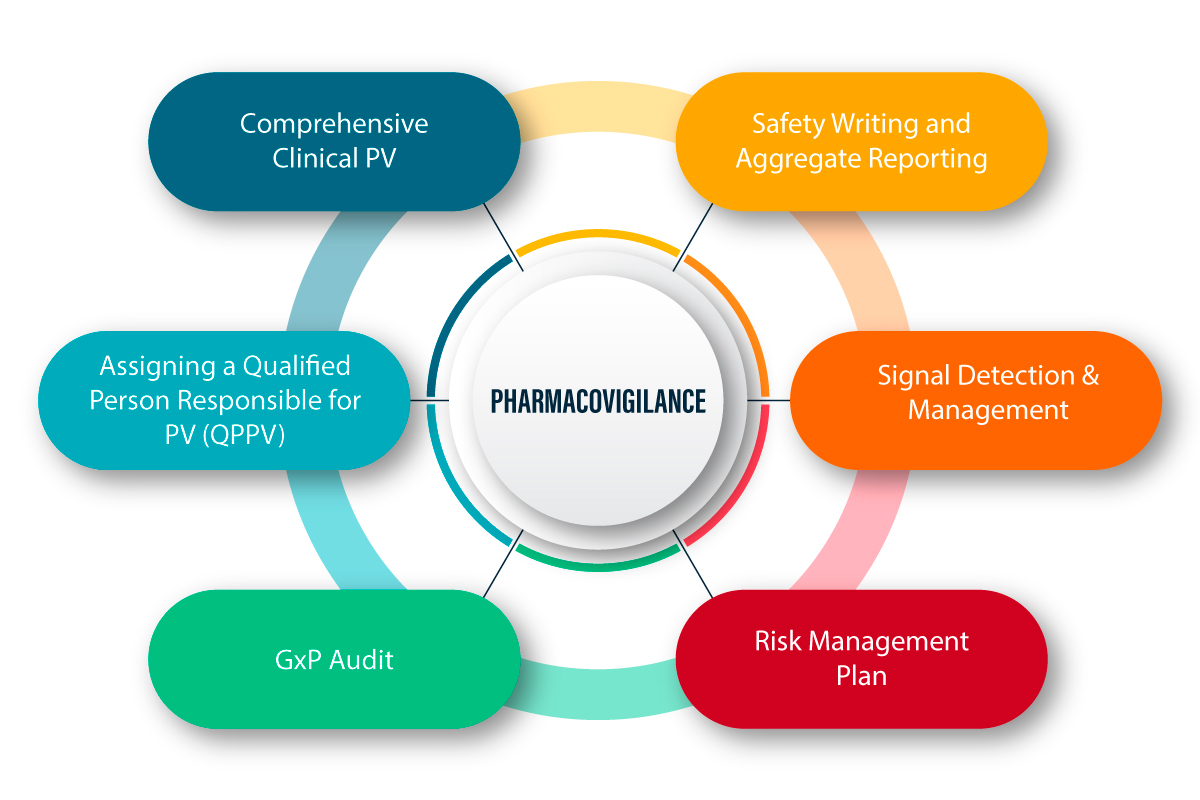
Let’s be real: in today’s business world, you can’t just ignore compliance. It’s not a box to tick—it’s something you’ve…
Read MoreHighlights and benefits of the recent U.S.–India trade agreement, with a focus on what it does (including tariff reductions) and…
Read MoreIndia-EU Free Trade Agreement — The “Mother of All Deals” Big news on the trade front: On January 27, 2026,…
Read More
Noida’s business scene is changing fast, and if you want your products to hit the market without hiccups, you need…
Read MoreLooking for Drug & Medical Device Registration in Noida? Get Full Regulatory Support from Experts Noida’s become a go-to spot…
Read MoreLooking for Drug & Medical Device Registration in Noida? Get Full Regulatory Support from Experts Noida’s become a go-to spot…
Read More